MEDIA
HELP
The CARA Platform from Generis is a powerful, highly configurable platform built for you to manage your enterprise content, data, as well as business processes efficiently. As an official Generis Certified Partner, our content migration software migration-center supports both CARA for OpenText Documentum to the new cloud-based CARA Platform. We simplify migrations to the CARA Platform environment by featuring flexible ways to upload and transform content, import documents, content-less objects, structures, and audit trails.
In order to explore how the combination of the CARA Platform and migration-center can maximize value for customers, we are pleased to talk with Karo Rogowska, Marketing Manager at Generis.
Many of our clients choose the CARA Platform as their go-to ECM solution as it is designed to specifically address the challenges within strictly regulated markets such as the life sciences and financial sector. What capabilities make CARA a compliant platform?
Karo: CARA was designed particularly for highly regulated industries with ever-shifting compliance requirements, so compliance and security are at the core of our platform. Functionality like multi-factor authentication and SSO control access to the platform, while fully configurable security rules based down to the individual role, group, user, and even document ensure users can access everything they need to do their job and no more.
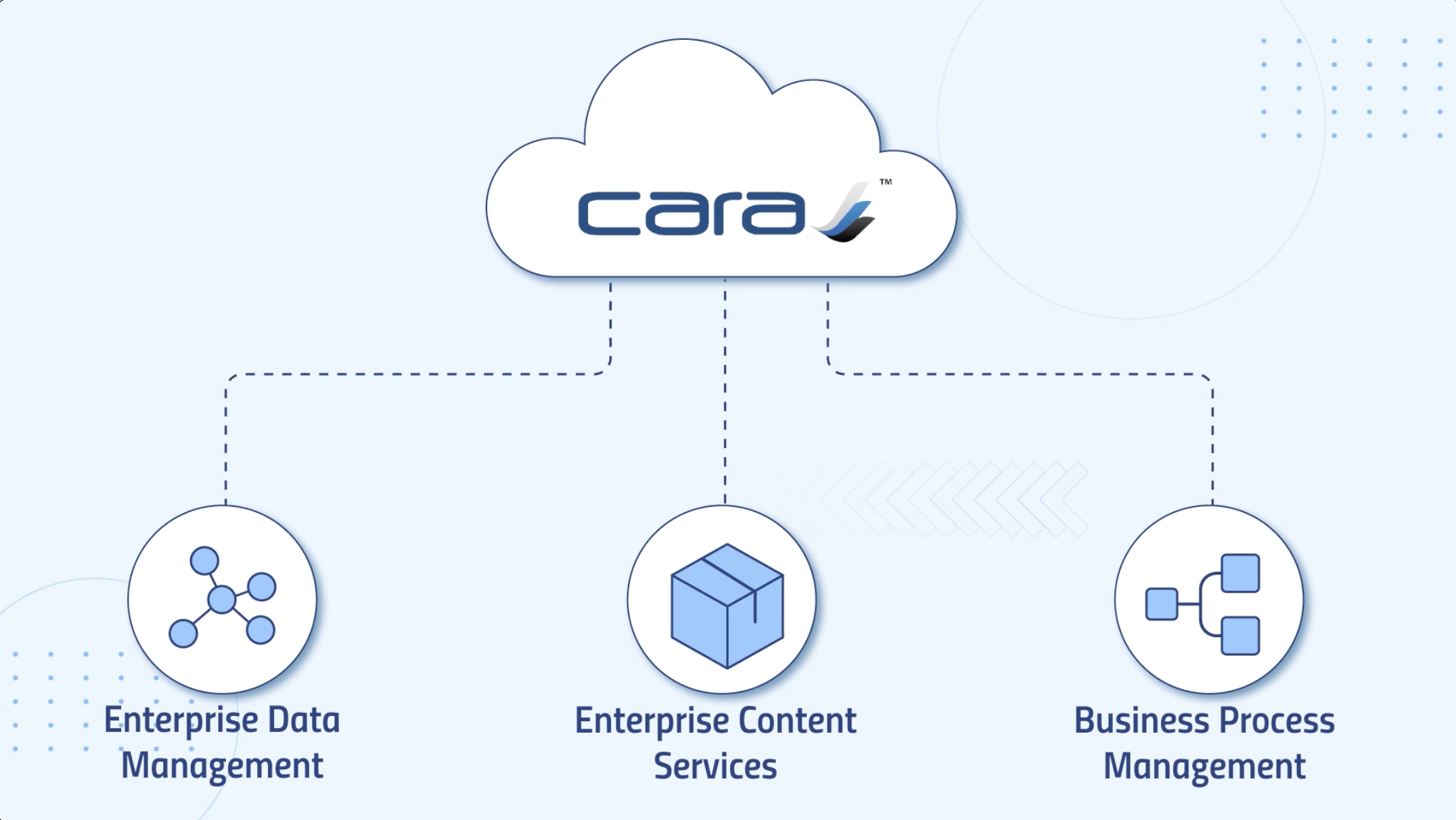
CARA is Generis’ unified data, content, and business process management platform for regulated industries.
Audits usually create lots of extra work and stress if the enterprise information is not properly managed. With CARA, everything is auditable, from individual data value and document changes, to user activity, workflows, versioning, and lifecycles. All of this helps organizations remain compliant across the enterprise and meet industry standards like: GDPR, HIPAA, DICOM, GxP, 21 CFR Part 11, EU Annex 11, SOX, FCPA, and other global requirements.
In the past, CARA was deployed as an OpenText Documentum business rules layer. Lately we have seen a shift in your strategy towards a broader content services platform. What can you tell us about the new CARA Platform, and why should users work with it?
Karo: We started working on the new CARA Platform when we first saw the needs of our customers shifting towards more modern ways of working, without the restrictions of having another repository underneath. We wanted to create a solution which would make the monotonous daily tasks around documentation and data easier and more efficient.
From this we then created the CARA Life Sciences Platform, with out-of-the-box solutions across Quality, Regulatory, Safety, and Clinical for Life Sciences organizations, all from the same platform interface. CARA users can save time and effort by moving seamlessly through configurable workspaces: data and content can flow between any solution on the platform without integration, enabling collaboration, discovery, and traceability across the business. We eliminate the need for a network of siloed systems.
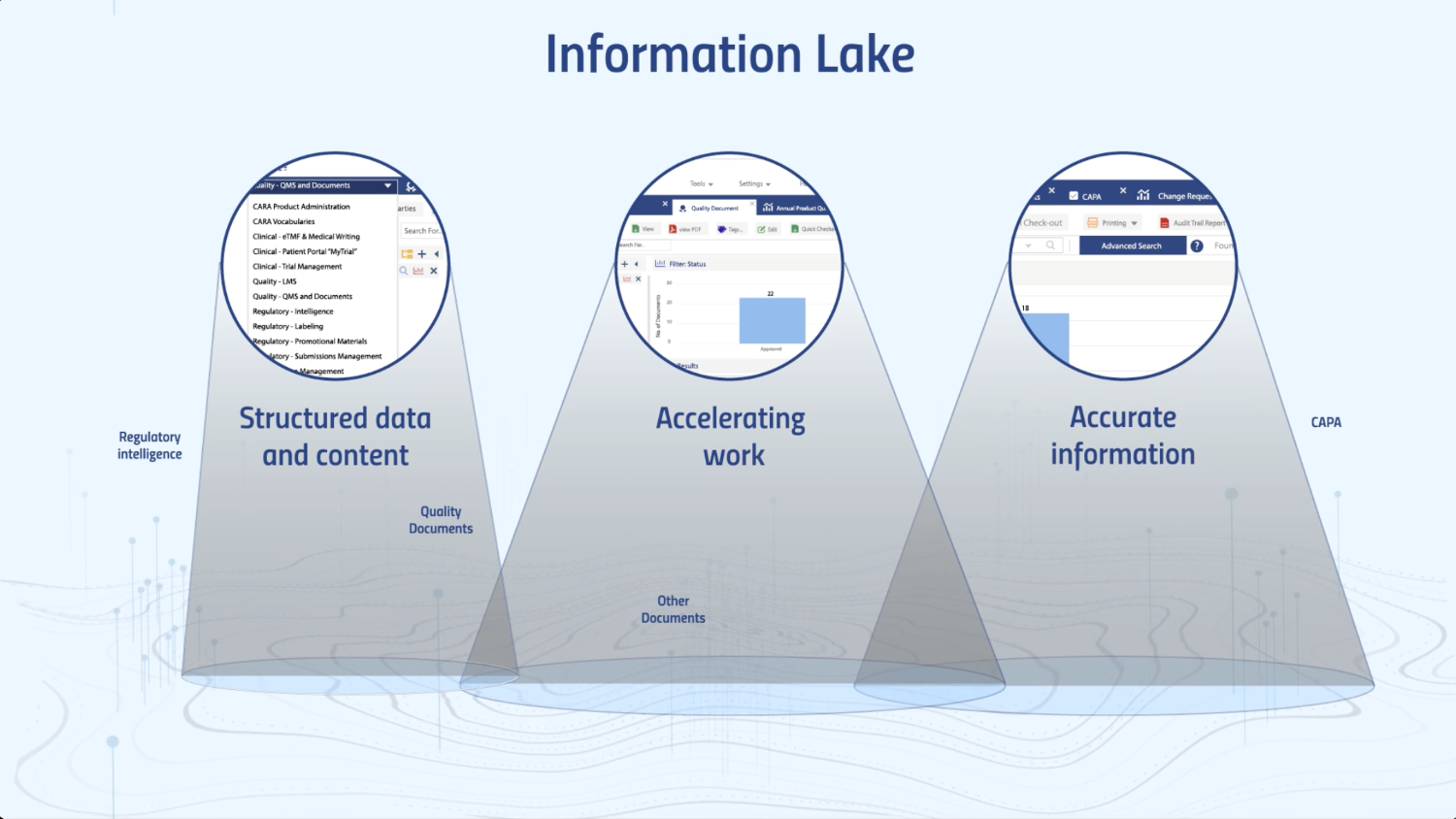
Users can create, locate, and reuse information instantly in any process across the organization thanks to the underlying single information lake.
What really differentiates us from other content services platform is our strong focus on the data, all the information is located in the underlying structured data lake which enables discovery through its ease of information retrieval (using powerful search to return millions of documents in under a second), generating analysis reports for insights, and allowing users to gain oversight of processes across the organisation without ever leaving the platform.
Considering the vital role of compliance in highly regulated frameworks, migration-center provides a number of functionalities that enable a smooth and secure onboarding process to the CARA Platform. What is your experience with our end-to-end software?
Karo: With our partnership, customers benefit from the expertise and the deep industry knowledge from both of our companies. fme understands the challenges and potential difficulties when it comes to migration and can prevent these from happening. Proper migration practices can make the process much less disruptive for the wider company which in turn helps with overall user adoption and user experience. Due to our longstanding collaboration, the fme teams know CARA inside out, they work as an extension of our experts and can help guide companies through their journeys.
When it comes to migration-center’s platform-specific capabilities, its CARA connector ensures a secure migration of RIM data such as document version trees, relationships, audit trails, and structures from various source applications and/or file shares into Generis CARA5 systems.
Another strength of migration-center is its flexibility: it offers the ability to re-run a migration on the same set of objects multiple times. Especially when you have to quickly adjust to changing requirements of projects and in case of delta migrations, this is very useful.
We have also experienced the flexibility of the product in terms of the approach to document content migration. Various options allow for greater time savings during the scanning and importing phases of the migration. On top of that, migration-center is capable of performing migrations to previous Documentum-based, pre-CARA5 systems using the Documentum connector.
A closer look at the CARA Platform shows that the best-of-breed applications, along with its process automation and rich features, provide a powerful, fully compliant solution for regulated business processes.
If you plan to replace your legacy applications with the CARA Platform, please don’t hesitate to contact us. Gathering 20+ years of experience in content migration projects for pharmaceutical clients, our team of certified experts is happy to help you analyze your current usage and achieve a faster implementation while maintaining high quality results and cost efficiency.

Get in touch with us today to find out how we can assist you!

 Accelerating migrations to Hyland Alfresco & Nuxeo content management ...
Accelerating migrations to Hyland Alfresco & Nuxeo content management ...